Clinical Trials for First-Line
Therapies in NSCLC
PD-L1 High ≥50%
Preferred therapy for programmed death-ligand 1 (PD-L1) high advanced/metastatic non-small cell lung cancer (mNSCLC) includes the anti-programmed cell death protein 1 (PD-1) monoclonal antibodies pembrolizumab or cemiplimab as monotherapy or in combination with histology-based doublet chemotherapy, or monotherapy with the anti-PD-L1 monoclonal antibody atezolizumab (Table 1).1
Immune Checkpoint Inhibitor Monotherapy
The efficacy and safety of single-agent anti-PD-1 or -PD-L1 (PD-[L]1) therapy versus chemotherapy for mNSCLC with PD-L1 expression ≥50% were evaluated in four phase III studies:
- KEYNOTE-024 (N = 305)2,3
- KEYNOTE-042 (N = 1274)4
- EMPOWER-Lung 1 (N = 563)5-7
- IMpower110 (N = 205)8,9
In all four studies, single-agent anti-PD(L)1 therapy significantly improved progression-free survival (PFS) and overall survival (OS) compared with chemotherapy.
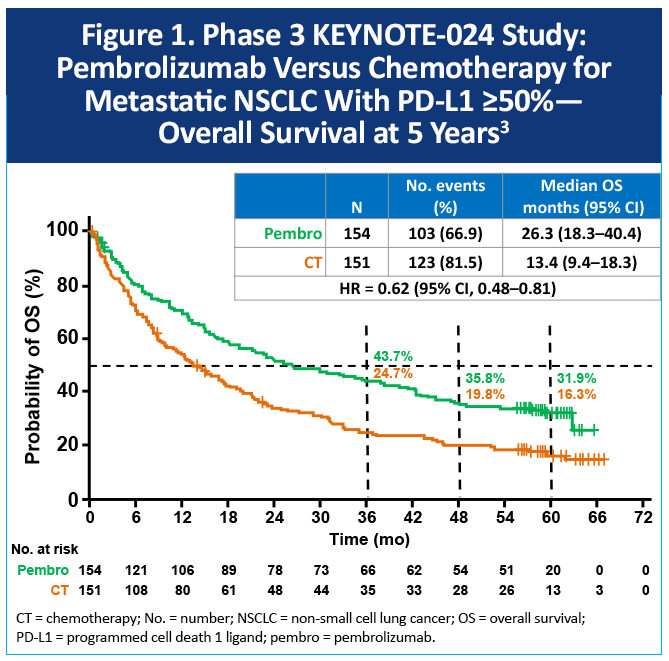
KEYNOTE-024 (non-squamous and squamous)
KEYNOTE-024 enrolled patients with mNSCLC that was non-squamous or squamous histology, PD-L1 tumor proportion score (TPS) ≥50%, and non-EGFR or –ALK altered. Participants (N = 305) were randomized to receive pembrolizumab or platinum-based chemotherapy.2 As shown in Figure 1, at a median follow up of 60 months, the median OS was 26.3 months with pembrolizumab and 13.4 months with chemotherapy (hazard ratio [HR], 0.62; 95% confidence interval [CI], 0.48-0.81). Five-year OS rates were 32% and 16%, respectively.3 Of note, among patients initially assigned to chemotherapy, 66% received subsequent anti–PD-1 or PD-L1 therapy.
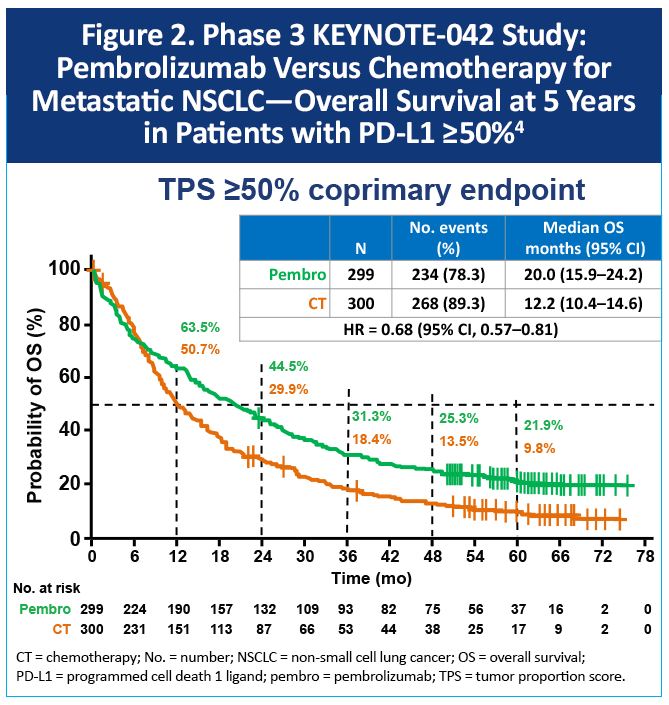
KEYNOTE-042 (non-squamous and squamous)
The phase 3 KEYNOTE-042 study evaluated pembrolizumab versus chemotherapy for patients with locally advanced/metastatic, non-squamous or squamous NSCLC without EGFR/ALK alterations and with PD-L1 TPS ≥1%.4 Half of the patients in the study had PD-L1 expression ≥50%. Among these patients, the median OS was 20.0 months with pembrolizumab and 12.2 months with chemotherapy (HR, 0.68; 95% CI, 0.57-0.81), Figure 2. The 5-year OS rates were 22% versus 10% respectively.
EMPOWER-Lung 1 (non-squamous and squamous)
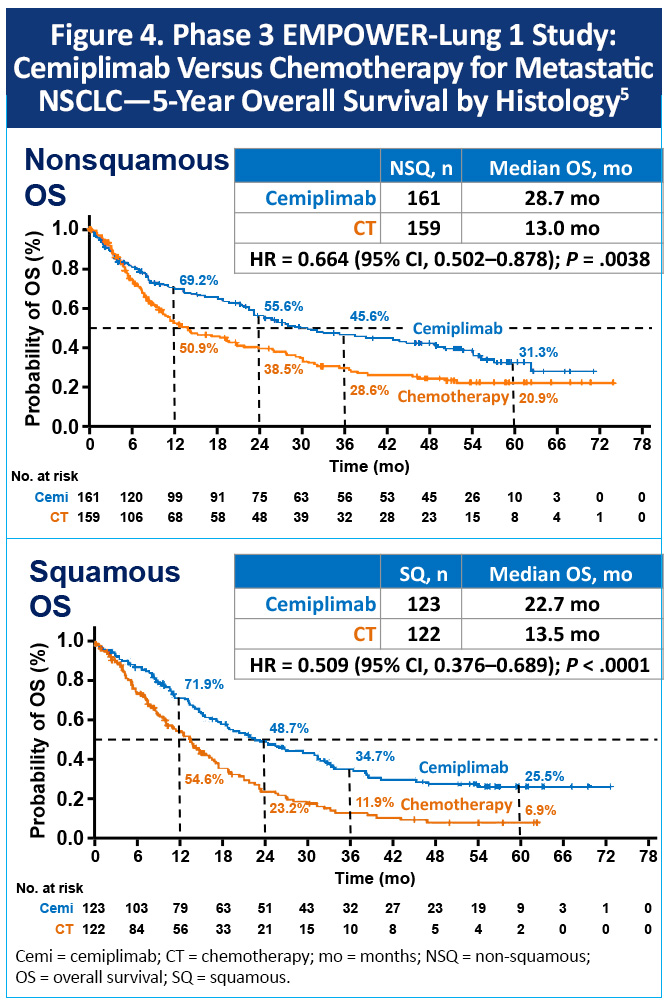
The phase 3 EMPOWER-Lung 1 trial enrolled patients with advanced NSCLC with PD-L1 expression ≥50% and without EGFR, ALK, or ROS1 aberrations. The study randomized 712 patients to cemiplimab or chemotherapy. At a median follow-up of 59.6 months, the median OS was 26.1 months with cemiplimab and 13.3 months with chemotherapy (HR, 0.59; 95% CI, 0.48–0.72), Figure 3.5 The five-year OS rates were 29.0% versus 15.0%, respectively. Survival benefits were observed regardless of histology (Figure 4).
IMpower110 (non-squamous or squamous)
Histology-based doublet chemotherapy plus immune checkpoint inhibition
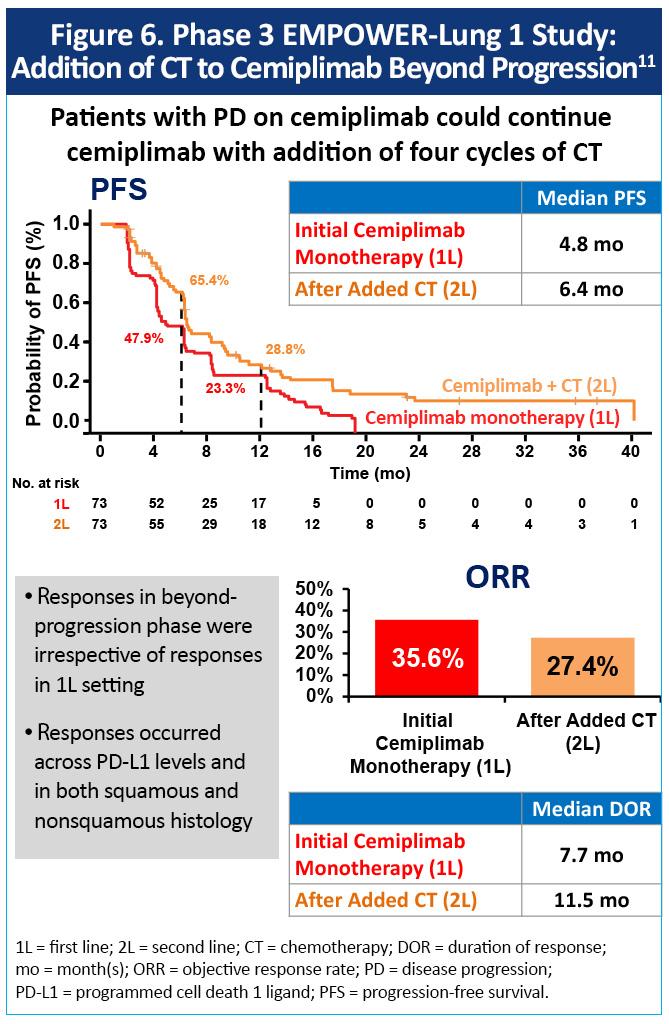
Histology-based doublet chemotherapy plus pembrolizumab or cemiplimab has not been directly compared with anti–PD-1 monotherapy in mNSCLC with PD-L1 ≥50%. However, per expert consensus, escalation with chemotherapy may be considered for patients with a significant tumor burden or in need of a rapid response to therapy (eg, high risk of visceral crisis).10 In EMPOWER-Lung 1, patients with disease progression while on cemiplimab could continue cemiplimab with the addition of 4 cycles of chemotherapy.11 The overall response rate to cemiplimab beyond progression was 27% (vs 36% response to initial cemiplimab monotherapy), the median duration of overall response was 11.5 months (vs 7.7 months), and the median PFS was 6.4 months (vs 4.8 months), Figure 6. Responses in the beyond-progression phase were irrespective of responses in the first-line setting, PD-L1 levels, or squamous versus nonsquamous histology.
Ongoing studies are underway to develop biomarkers that identify patients who would benefit from escalation of treatment (eg, mutations in STK11/KEAP1).10 Additionally, the ongoing phase III Eastern Cooperative Oncology Group (ECOG)–American College of Radiology Imaging Network (ACRIN) INSIGNA trial (NCT03793179) is evaluating first-line immunotherapy alone or in combination with chemotherapy in induction/maintenance or post-progression for non-squamous mNSCLC to aid more personalized care in the future.
PD-L1 ≥1% to 49%
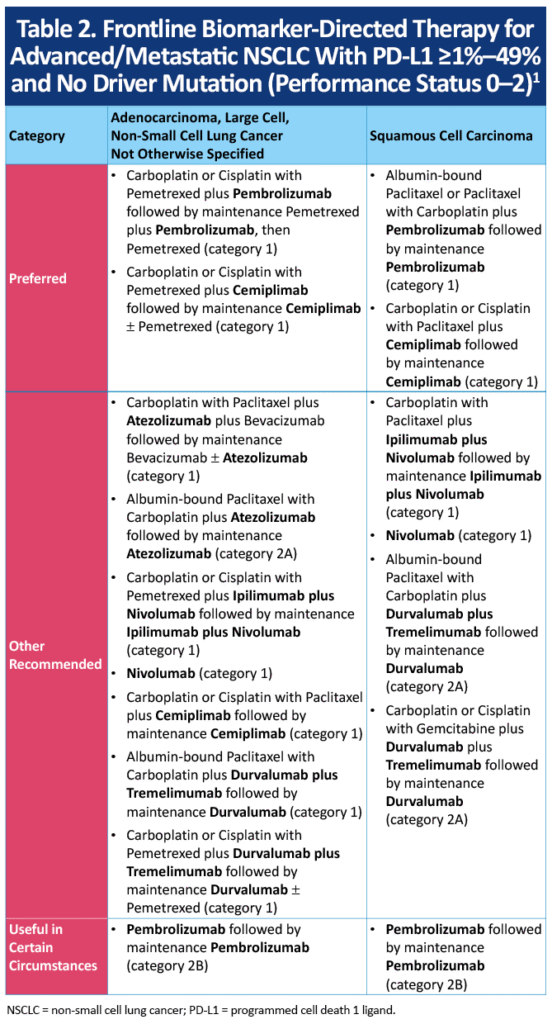
Preferred first-line therapy for mNSCLC with PD-L1 expression 1% to 49% and no driver mutation is pembrolizumab or cemiplimab in combination with histology-based doublet chemotherapy (Table 2).1
Histology-based doublet chemotherapy plus immune checkpoint inhibition
KEYNOTE-189 (non-squamous)
The phase 3 KEYNOTE-189 study evaluated pembrolizumab plus standard chemotherapy (pemetrexed plus a platinum agent) versus standard chemotherapy for non-squamous mNSCLC without EGFR or ALK mutations.12 Long-term follow-up from this study demonstrated that among patients with PD-L1 tumor expression of 1% to 49%, 5-year OS was approximately 20% versus 8%, with a median OS of 22 months versus 12 months (Figure 7).13
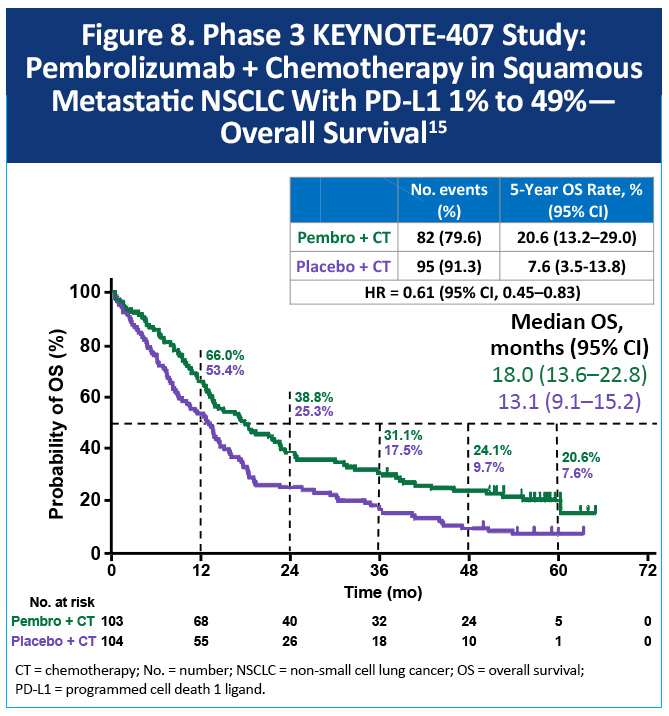
KEYNOTE-407 (squamous)
The phase 3 KEYNOTE-407 study randomized 559 patients with previously untreated, squamous mNSCLC to receive carboplatin and paclitaxel/nab-paclitaxel with pembrolizumab or placebo.14 At a median follow-up of 56.9 months, the estimated 5-year OS rates were 18.4% with pembrolizumab plus chemotherapy and 9.7% with chemotherapy (HR, 0.62; 95% CI, 0.52-0.74).15 Among patients with PD-L1 1% to 49% expression, the median OS was 18.0 months versus 13.1 months, respectively (HR, 0.61; 95% CI, 0.45-0.83), with 5-year OS rates of 20.6% and 7.6%, respectively (Figure 8).
EMPOWER-Lung 3 (non-squamous and squamous)
The EMPOWER-Lung 3 study enrolled patients with squamous or non-squamous mNSCLC of any PD-L1 expression level that was not EGFR, ALK, or ROS1 altered (Figure 9).16-18 At 5 years of follow-up (median 61 months), the final median OS in the overall population was 21.1 months with cemiplimab plus chemotherapy and 12.9 months with chemotherapy alone (HR, 0.66; 95% CI, 0.51–0.83; P =.0002).17 Looking at OS by histology, the median OS for 200 patients with squamous NSCLC was 22.3 months versus 13.8 months (HR, 0.68; 95% CI, 0.49–0.94), Figure 10. For patients with nonsquamous NSCLC (n = 266), median OS was 19.4 months versus 12.4 months (HR, 0.62; 95% CI, 0.46–0.82), Figure 11. In the PD-L1 TPS 1% to 49% subgroup (non-squamous and squamous), median OS was 23 months versus 12 months, respectively (HR, 0.46; 95% CI, 0.32-0.66) at 2 years of follow up.
Unknown or Negative PD-L1 Expression, TPS 0%
Preferred therapy for PD-L1 expression <1% is also pembrolizumab or cemiplimab with histology-based doublet chemotherapy (Table 3).1
Histology-based doublet chemotherapy plus immune checkpoint inhibition
KEYNOTE-189 (non-squamous)
At 5 years of follow up in the KEYNOTE-189 study, the median OS among patients with nonsquamous mNSCLC and PD-L1 <1% was 17 months with pembrolizumab plus chemotherapy (n = 127) versus 10.2 months with chemotherapy alone (n = 63) with an HR of 0.55 (95% CI, 0.39 to 0.76). The 5-year OS rates were 10% and 5%, respectively.13 Consistent findings were reported from an exploratory pooled analysis of 442 patients with previously untreated mNSCLC with PD-L1 expression <1% who participated in phase 3 studies of pembrolizumab plus chemotherapy versus placebo plus chemotherapy.19 The analysis included data from the KEYNOTE-189 global (NCT02578680) and Japan extension (NCT03950674) studies of metastatic nonsquamous NSCLC without EGFR or ALK alterations and the KEYNOTE-407 global (NCT02775435) and People’s Republic of China extension (NCT03875092) studies of metastatic squamous NSCLC.
EMPOWER-Lung 3 (non-squamous or squamous)
At 2 years of follow up in the EMPOWER-Lung 3 study, the median OS among patients with advanced NSCLC (non-squamous and squamous) with PD-L1 <1% was 13 months with cemiplimab plus chemotherapy versus 14.2 months with chemotherapy (HR, 0.73; 95% CI, 0.50 to 1.08).18 In patients with PD-L1 <1% and squamous histology, the median OS was 21.9 months versus 16.7 months (HR, 0.60; 95% CI, 0.30-1.20). In the PD-L1 <1%, non-squamous histology subgroup, the median OS was 9.6 months versus 13.0 months (HR, 1.26; 0.74-2.12).
Patient Selection for Immune Checkpoint Inhibition10
Preferred first-line therapy for patients with advanced NSCLC and no driver mutation is an immune checkpoint inhibitor (ICI)-based regimen. The decision to use an ICI in combination with chemotherapy is largely based on PD-L1 expression level, although other factors may influence decision making. Chemotherapy is typically cisplatin or carboplatin in combination with pemetrexed for non-squamous tumors and paclitaxel for squamous tumors. Patients who are not candidates for an ICI (eg, active autoimmune disease, solid organ transplant, or history of severe immune-related adverse events) should receive doublet chemotherapy. It is important for clinicians to understand that patients with pre-existing, non-neurologic, non-life-threatening, controlled autoimmune conditions may be eligible for an ICI under close supervision of a multidisciplinary team. Non-platinum combinations may be considered for patients with a contraindication to platinum therapy (eg, gemcitabine plus docetaxel, gemcitabine plus vinorelbine).
References
- National Comprehensive Cancer Network® NCCN Clinical Practice Guidelines in Oncology. Non-Small Cell Lung Cancer. Version 5.2026. March 13, 2026. https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1450
- Reck M, Rodríguez-Abreu D, Robinson AG, et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med. 2016;375:1823-1833. https://doi.org/10.1056/NEJMoa1606774
- Reck M, Rodríguez-Abreu D, Robinson AG, et al. Five-year outcomes with pembrolizumab versus chemotherapy for metastatic non-small-cell lung cancer with PD-L1 tumor proportion score ≥ 50. J Clin Oncol. 2021;39:2339-2349. https://doi.org/10.1200/jco.21.00174
- de Castro G, Jr, Kudaba I, Wu YL, et al. Five-year outcomes with pembrolizumab versus chemotherapy as first-line therapy in patients with non-small-cell lung cancer and programmed death ligand-1 tumor proportion score ≥ 1% in the KEYNOTE-042 study. J Clin Oncol. 2023;41:1986-1991. https://doi.org/10.1200/JCO.21.02885
- Kilickap S, Baramidze A, Sezer A, et al. Cemiplimab monotherapy for first-line treatment of patients with advanced NSCLC with PD-L1 expression of 50% or higher: Five-year outcomes of EMPOWER-Lung 1. J Thorac Oncol. 2025;20:941-954. https://doi.org/10.1016/j.jtho.2025.03.033
- Sezer A, Kilickap S, Gümüş M, et al. Cemiplimab monotherapy for first-line treatment of advanced non-small-cell lung cancer with PD-L1 of at least 50%: A multicentre, open-label, global, phase 3, randomised, controlled trial. Lancet. 2021;397:592-604. https://doi.org/10.1016/s0140-6736(21)00228-2
- Özgüroğlu M, Kilickap S, Sezer A, et al. First-line cemiplimab monotherapy and continued cemiplimab beyond progression plus chemotherapy for advanced non-small-cell lung cancer with PD-L1 50% or more (EMPOWER-Lung 1): 35-month follow-up from a mutlicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2023;24:989-1001.
https://doi.org/10.1016/s1470-2045(23)00329-7 - Herbst RS, Giaccone G, de Marinis F, et al. Atezolizumab for first-line treatment of PD-L1-Selected patients with NSCLC. N Engl J Med. 2020;383:1328-1339. https://doi.org/10.1056/nejmoa1917346
- Jassem J, de Marinis F, Giaccone G, et al. Updated overall survival analysis from IMpower110: Atezolizumab versus platinum-based chemotherapy in treatment-naive programmed death-ligand 1-selected NSCLC. J Thorac Oncol. 2021;16:1872-1882. https://doi.org/10.1016/j.jtho.2021.06.019
- Reuss JE, Bazhenova L, Ismaila N, et al. Therapy for stage iv non–small cell lung cancer without driver alterations: ASCO living guideline, 2026.3.0. J Clin Oncol. 2026;44:e56-e88. https://doi.org/10.1200/JCO-25-02825
- Garassino MC, Baramidze A, Kilickap S, et al. 16P: Continued cemiplimab with addition of chemotherapy beyond progression in patients with advanced NSCLC on 1L cemiplimab monotherapy: 5-year outcomes of EMPOWER-Lung 1. J Thoracic Oncol. 2025;20:(3 suppl 1):S19-S21. https://doi.org/10.1016/S1556-0864(25)00211-4
- Gandhi L, Rodríguez-Abreu D, Gadgeel S, et al. Pembrolizumab plus chemotherapy in metastatic non-small-cell lung cancer. N Engl J Med. 2018;378:2078-2092. https://doi.org/10.1056/NEJMoa1801005
- Garassino MC, Gadgeel S, Speranza G, et al. Pembrolizumab plus pemetrexed and platinum in nonsquamous non-small-cell lung cancer: 5-year outcomes from the phase 3 KEYNOTE-189 study. J Clin Oncol. 2023;41:1992-1998. https://doi.org/10.1200/JCO.22.01989
- Paz-Ares L, Luft A, Vicente D, et al. Pembrolizumab plus chemotherapy for squamous non–small-cell lung cancer. N Engl J Med. 2018;379:2040-2051. https://doi.org/10.1056/NEJMoa1810865
- Novello S, Kowalski DM, Luft A, et al. Pembrolizumab plus chemotherapy in squamous non–small-cell lung cancer: 5-year update of the phase III KEYNOTE-407 study. J Clin Oncol. 2023;41:1999-2006. https://doi.org/10.1200/JCO.22.01990
- Gogishvili M, Melkadze T, Makharadze T, et al. Cemiplimab plus chemotherapy versus chemotherapy alone in non-small cell lung cancer: A randomized, controlled, double-blind phase 3 trial. Nat Med. 2022;28:2374-2380. https://doi.org/10.1038/s41591-022-01977-y
- Baramidze A, Makharadze T, Gogishvili M, et al. Cemiplimab plus chemotherapy vs chemotherapy in advanced NSCLC: 5-year results from phase 3 EMPOWER-Lung 3 Part 2 trial. Presented at: 2025 IASLC World Conference on Lung Cancer; September 6-10, 2025; Barcelona, Spain. Oral presentation MA10.09.
- Makharadze T, Gogishvili M, Melkadze T, et al. Cemiplimab plus chemotherapy versus chemotherapy alone in advanced NSCLC: 2-year follow-up from the phase 3 EMPOWER-lung 3 part 2 trial. J Thorac Oncol. 2023;18:755-768. https://doi.org/10.1016/j.jtho.2023.03.008
- Gadgeel SM, Rodriguea-Abreu D, Halmos B, et al. Pembrolizumab plus chemotherapy for metastatic NSCLC with programmed cell death ligand 1 tumor proportion score less than 1%: pooled analysis of outcomes after five years of follow-up. J Thorac Oncol. 2024;19:1228-1241. https://doi.org/10.1016/j.jtho.2024.04.011
All URLs accessed April 29, 2026