Biomarker-Directed Use of Immune Checkpoint Inhibitors for NSCLC
Immune checkpoint inhibitors (ICI), particularly those targeting programmed cell death protein 1 (PD-1) or programmed cell death-ligand 1 (PD-L1), have demonstrated durable efficacy in a subset of patients with non-small cell lung cancer (NSCLC), establishing themselves as the cornerstone of first-line therapy for non-driver mutated NSCLC.1,2
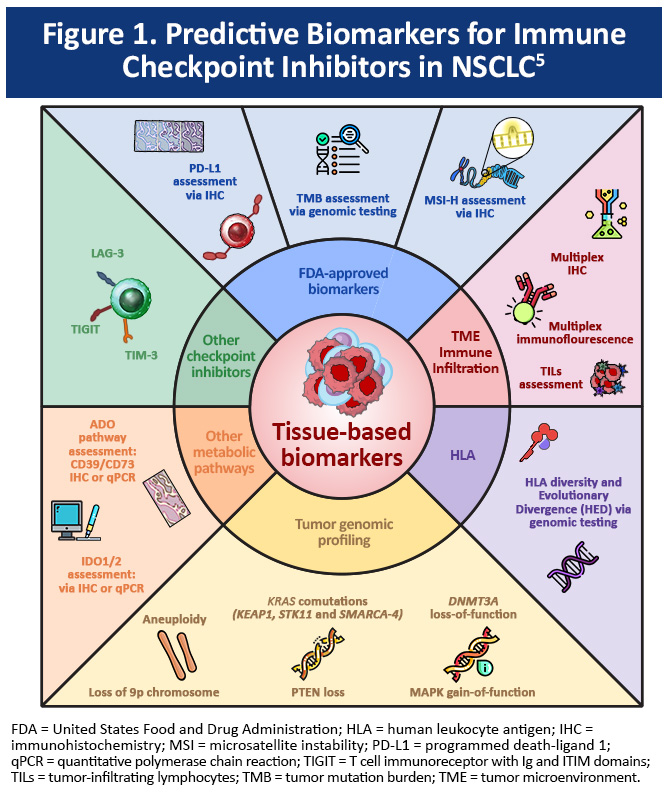
Interpretation of biomarker data remains an important priority for medical oncologists managing patients with advanced NSCLC.1-5 Predictive biomarkers of ICI response currently approved by the United States FDA (shown in Figure 1) are tumor PD-L1 expression, tumor mutation burden (TMB), and microsatellite-instability (MSI).3 Current evidence-based guidelines include PD-L1 testing as part of a broader biomarker testing strategy for all patients diagnosed with NSCLC.1,2 Importantly, some oncogenic drivers (ie, EGFR exon 19 deletion or L858R point mutation; ALK, RET, or ROS1 rearrangements) are associated with less benefit from anti-PD-L1 therapy.1
Practice Tips
- Patients should be tested for these drivers prior to initiating anti-PD-L1 therapy
- Targeted therapy should take priority over immunotherapy for these patients
- Recognizing the importance of tumor proportion score (TPS) of PD-L1 positivity and TMB is crucial in the selection of appropriate therapy
Existing biomarkers are imperfect; therefore, research is underway to identify predictive biomarkers beyond tumor PD-L1 expression to aid patient selection for ICI therapy (Figure 1).3-5
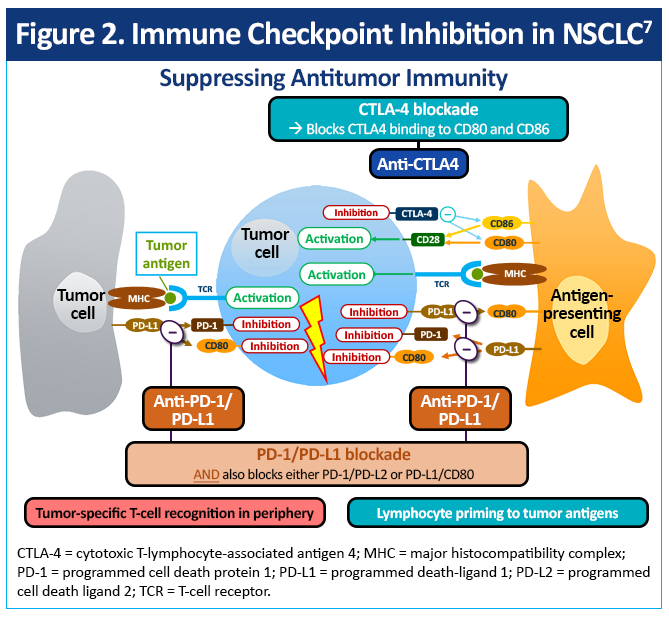
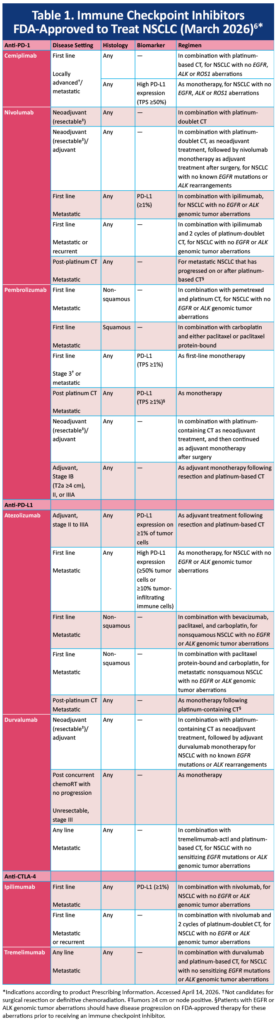
Immune checkpoint inhibitors approved to treat NSCLC include those that target cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4), PD-1 or PD-L1 (Figure 2).1,2,6,7 These agents carry varying indications as shown in Table 1.1,2
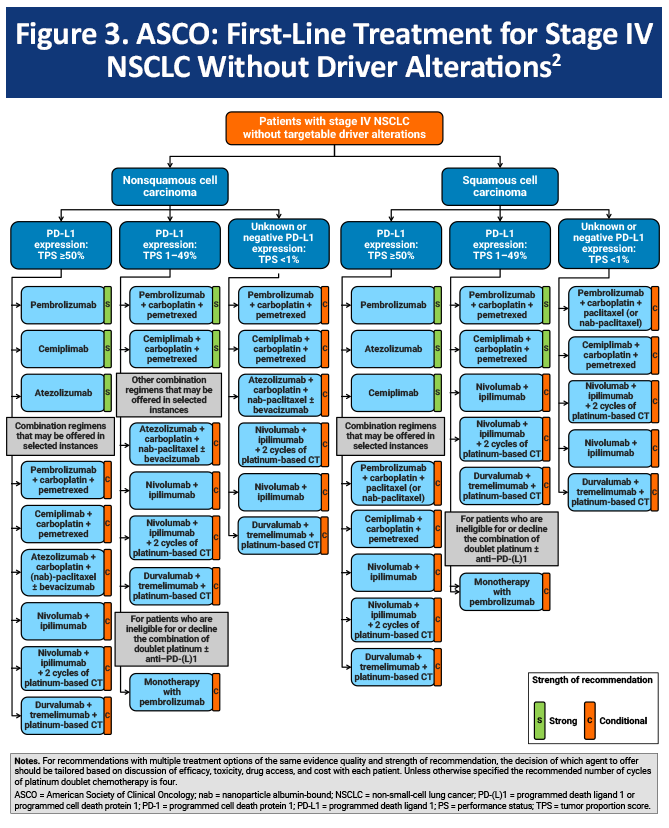
Immune checkpoint inhibition-based therapy is the first-line standard of care for patients with metastatic NSCLC that does not harbor actionable mutations (Figure 3).2 Regimen selection is based on various factors including PD-L1 expression level, histology, disease symptoms and burden, and toxicity concerns.
References
- NCCN Clinical Practice Guidelines in Oncology®. NCCN Clinical Practice Guidelines in Oncology. Non-Small Cell Lung Cancer. Version 5.2026. https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf
- Reuss JE, Bazhenova L, Ismaila N, et al. Therapy for stage IV non–small cell lung cancer without driver alterations: ASCO living guideline, 2026.3.0. J Clin Oncol. 2026;44:e56-e88. https://doi.org/10.1200/JCO-25-02825
- Wang SL, Chan TA. Navigating established and emerging biomarkers for immune checkpoint inhibitor therapy. Cancer Cell. 2025;43:641-664. http://doi.org/10.1016/j.ccell.2025.03.006
- Yamaguchi H, Hsu JM, Sun L, Wang SC, Hung MC. Advances and prospects of biomarkers for immune checkpoint inhibitors. Cell Rep Med. 2024;5:101621. https://doi.org/10.1016/j.xcrm.2024.101621
- Garriazzo E, Colamartini F, Ubaldi M, Santo V, Brunetti L, Tomarelli C. Emerging predictive biomarkers of immunotherapy sensitivity in patients with non-small cell lung cancer. Immunotargets Ther. 2025;15:567238. https://doi.org/10.2147/ITT.S567238
- National Cancer Institute. Drugs Approved for Lung Cancer. Updated March 14, 2025. https://www.cancer.gov/about-cancer/treatment/drugs/lung
- Suraya R, Tachihara M, Nagano T, Nishimura Y, Kobayashi K. Immunotherapy in advanced non-small cell lung cancers: Current status and updates. Cancer Manag Res. 2022;14:2079-2090. https://doi.org/10.2147/CMAR.S366738
All URLs accessed April 26, 2026